2023 Study: Adipose Therapy Offers 3-Year Relief in 80% of Knee Osteoarthritis (OA) Patients
Date: 6 May 2026
Admin: Medical Affairs
The Question Every Patient Asks First
When patients consider any treatment for knee osteoarthritis, one question almost always comes first: how long will this actually last?
It’s a fair question – and a financially important one. Cortisone shots wear off in six weeks (and damage the joint environment), hyaluronic acid can help for a season but not the next – you know the fatigue of treatments that offer relief on loan. You get a few good months, then the clock runs out, and you’re back at square one – stiffer, more cautious, and increasingly convinced that permanent limitation is just the price of a worn-out knee.
This pattern defines what we call the treatment gap – the space between conventional care that fades and surgery you’re not ready for, or don’t want. In that gap, many patients simply wait. They modify their activities, reduce their expectations, and manage. For a golfer, skier, or hiker in their 50s or 60s, that’s not a neutral outcome. That’s a slow loss of the life they’d planned.
A primary question about adipose therapy isn’t just does adipose therapy work. It’s how long does it work. Does the improvement hold at six months? A year? Two years? Three?
Your Own Biology as a Long-Term Strategy
Adipose tissue – the fat your body stores – contains a dense population of mesenchymal stem cells, fibroblasts, endothelial cells, extracellular matrix, and a milieu of various non-cell biological components. When harvested, mechanically processed, and injected directly into the joint, this tissue has the potential to directly treat the underlying causes of osteoarthritis. It doesn’t function like motor oil applied to a worn engine – lubricating while the underlying damage continues. Instead, it acts directly on the joint’s biology, potentially even reversing disease progression in ways that conventional injections do not.
That distinction matters when you’re asking about durability. A lubricant wears away. A biological process, if it takes hold, may have effects that persist.
A 2023 case series published in the Journal of Experimental Orthopaedics set out to document exactly how long those effects last. Researchers followed 49 patients (67 knees) for an average of nearly three years – with some individuals tracked for almost five – after a single intra-articular injection of micro-fragmented adipose tissue. All participants had already failed conventional conservative care and at least one prior intra-articular injection before enrolling. The results offer some of the most extended real-world durability data available on this treatment approach to date.
Learn More About Regenerative Matrix Therapy™
Study Overview: Three-Year Follow-Up of MFAT Injection for Recalcitrant Knee OA
Study Design: Retrospective case series, single-centre, no control group. Conducted at Humanitas Castelli Hospital, Bergamo, Italy. Level IV evidence.
Participants: Adults aged 18–80 with tibiofemoral knee OA at KL grade ≥ 1 who had already failed at least one conservative treatment (physical therapy, activity modification, or NSAIDs) and at least one prior intra-articular injection (corticosteroid, PRP, or hyaluronic acid). Patients with inflammatory arthritis, significant valgus/varus deformity (≥10°), or concurrent surgical procedures were excluded.
Sample Size: 49 patients (67 knees) enrolled. Full cohort followed to 6 months (n=67); 54 knees assessed at 12 months; 36 knees at 24 months; 25 knees at 36 months.
Severity Mix: KL grade 1 – 31.3% of knees; KL grade 2 – 61.2%; KL grade 3 – 7.5%. The majority of the cohort presented with moderate OA. The published KL distribution accounts for 100% of enrolled knees; no KL grade 4 knees appear in the reported breakdown. Per protocol, KL grade 4 patients were eligible only if they had declined surgical knee replacement.
Intervention: Single intra-articular injection of 10 mL autologous micro-fragmented adipose tissue (MFAT), harvested from the lower or lateral abdomen and processed using the Lipogems® closed-system mechanical device. No concurrent arthroscopic or surgical procedures were permitted. Post-injection rehabilitation: 5 days of unloading, progressive return to full weight-bearing over the following 5 days, proprioceptive exercises beginning at day 5.
Follow-Up: 1, 3, 6, 12, 24, and 36 months post-injection. Mean follow-up: 34.04 ± 13.62 months (range 11–59 months).
Endpoints: Primary – WOMAC score (pain, stiffness, and function subscales; total score) at all timepoints. Secondary – KOOS score (pain, symptoms, ADL, sport and recreation, and quality of life), and Minimal Clinically Important Difference (MCID) achievement rates for both instruments. Investigators also analyzed whether baseline severity, KL grade, or gender influenced outcomes.
Key Findings: A Single Injection, Still Working at Three Years
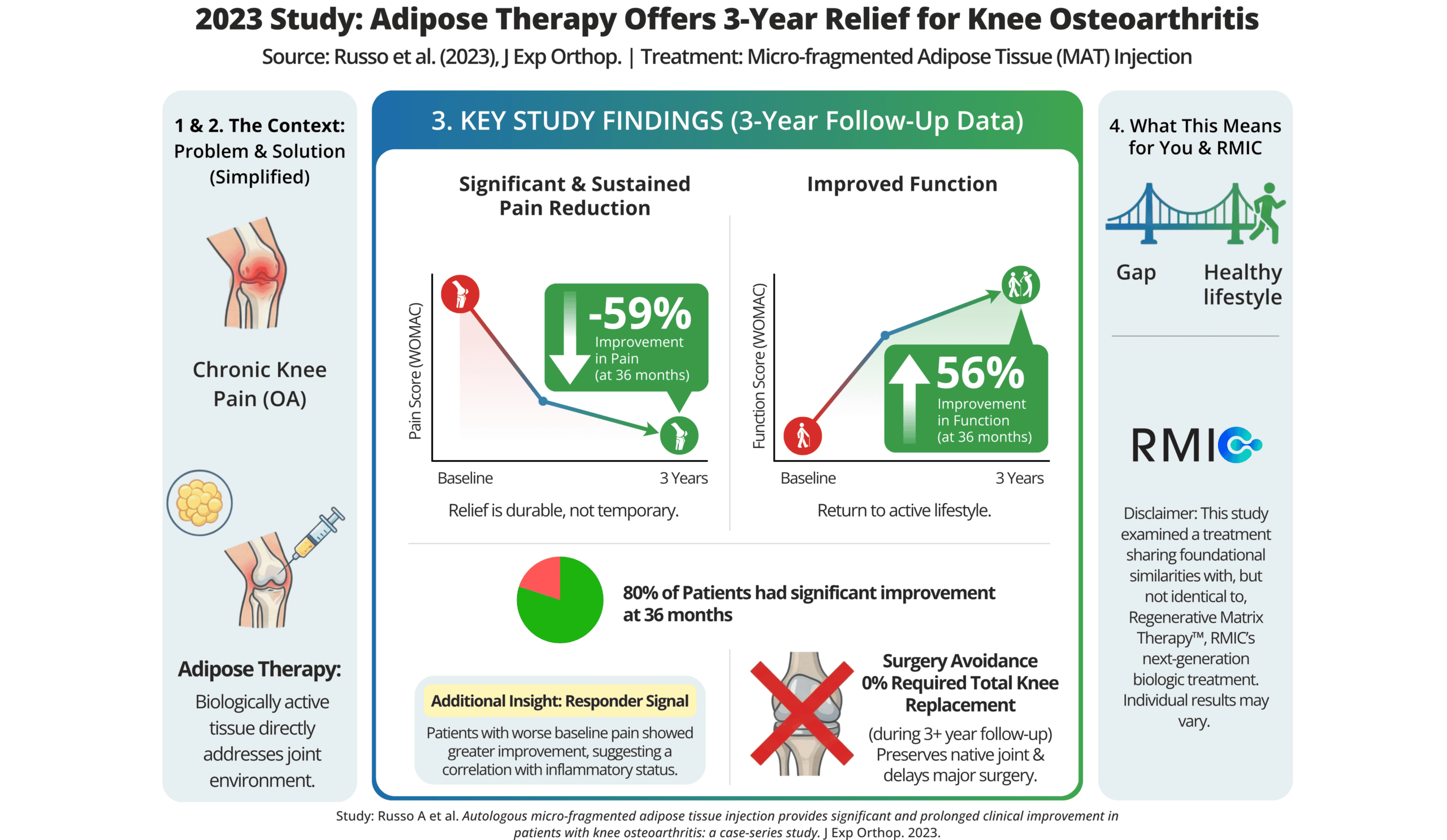
1. Pain dropped dramatically in months – and most of that reduction held at three years
WOMAC pain score fell from 10.73 at baseline to 3.15 at 6 months – a ~71% reduction (10.73 → 3.15; p<0.001). By 36 months, the score had risen modestly to 4.40, still representing a ~59% reduction from baseline (10.73 → 4.40; p<0.001). For most patients in the study, this translated from daily, activity-limiting pain to levels low enough to resume normal movement – and the majority of that relief was still measurable three years later.
2. Functional improvement was large, early, and remarkably stable
WOMAC function score – which captures capacity for everyday tasks like walking, climbing stairs, and rising from a chair – fell from 34.76 at baseline to 11.21 at 6 months (~68% improvement; 34.76 → 11.21; p<0.001). At 36 months, the score stood at 15.36, reflecting a ~56% improvement from baseline (34.76 → 15.36; p<0.001). For many patients, functional scores in this range represent the difference between independent, active daily life and the avoidance and accommodation that define uncontrolled OA.
3. Four in five patients maintained clinically meaningful functional benefit – at every single timepoint
The threshold for clinically meaningful functional improvement (WOMAC function MCID) was met by 76.1% of patients at 3 months, 79.1% at 6 months, 81.5% at 12 months, 83.3% at 24 months, and most notably, 80.0% at 36 months. What’s striking is not any individual figure – it’s the consistency. Across three years of follow-up, approximately four in five patients (80%) maintained improvement that exceeded the threshold for clinical meaningfulness, with no decay in that proportion over time.
4.Pain relief grew stronger with time – not weaker
The proportion of patients achieving clinically meaningful pain improvement (WOMAC pain MCID) actually increased as the follow-up extended: 28.3% at 3 months, rising steadily to 41.8% at 6 months, 46.3% at 12 months, 50.0% at 24 months, and 52.0% at 36 months. This counterintuitive trajectory – the opposite of what most patients expect from an injection – suggests that the biological response to MFAT may consolidate and mature over months to years, rather than peak and recede. By the three-year mark, more than half of patients had crossed the threshold for meaningful pain relief, and the trend continued upward throughout the observation window.
5. A comprehensive quality-of-life measure remained essentially unchanged from six months to three years
The KOOS composite score – integrating pain, symptoms, activities of daily living, sport and recreation, and quality of life – rose from 38.45 at baseline to 71.59 at six months, and stood at 71.09 at 36 months. Those near-identical values at six months and 36 months are among the study’s most compelling durability signals: the composite gains made in the first six months did not erode over the 30 months that followed. No patient in the study required knee replacement surgery during the entire follow-up period.
6. Safety profile was mild and consistently transient
The most common adverse event was temporary knee swelling and pain, occurring in 7 knees (10.4%), managed with cryotherapy, analgesics, and rest. Three patients (6%) experienced minor bruising at the harvest site, which resolved without intervention. No serious adverse events were recorded across the cohort.
What This Means for You
The question at the heart of this research is the one you’ve probably asked yourself: if I invest in this, will it still be working in a year? Two years? Three?
This study doesn’t guarantee a specific duration of effect – no ethical study can make that promise on behalf of an individual patient. But it offers something clinically meaningful: a documented pattern of stability. In a population of patients who had already failed conventional conservative care and prior injections, a single treatment produced improvements in pain and function that held for most participants through 24 months, remained substantially above baseline at 36 months, and – notably – showed an increasing proportion of patients achieving meaningful pain relief the longer they were followed. 80% of patients had meaningful relief that lasted at least 3 years.
That’s a different trajectory than what you’ve likely experienced with corticosteroids or hyaluronic acid.
While the study provides Level IV evidence, the data reveals significant patterns in which patients saw the most robust results. Rather than a uniform response, certain factors emerged as potential predictors of superior outcomes. For instance, female patients and those with lower-grade disease (KL grade 1–2) showed more robust outcomes. Most interestingly, patients starting with worse baseline symptoms were actually more likely to achieve meaningful clinical improvement. This suggests that the biological response from adipose-derived tissue may be particularly responsive to the more actively inflamed environments found in joints with higher symptomatic loads.
What this study can reasonably tell you is that the durability concern – the worry that any benefit will fade quickly, as prior treatments have – is not what the evidence shows for adipose therapy in this population.
Regenerative Matrix Therapy™ and RMIC
Regenerative Matrix Therapy™, offered by RMIC, shares foundational biological similarities with the micro-fragmented adipose tissue approach studied here. Both are designed to engage your own biology rather than introduce foreign substances into the joint. That said, Regenerative Matrix Therapy™ is not identical to the technique used in this specific study; RMIC’s proprietary Regenerative Matrix Therapy™ protocol has been reviewed by Health Canada with no objection.
If the question you’ve been carrying is how long can this realistically last, this research is directionally supportive – and it’s the kind of evidence we believe patients deserve to see.
Start Your Virtual Consultation
Research Highlights (For Clinicians)
Study Citation: Russo A, Cortina G, Condello V, Collarile M, Orlandi R, Gianoli R, Giuliani E, Madonna V. Autologous micro-fragmented adipose tissue injection provides significant and prolonged clinical improvement in patients with knee osteoarthritis: a case-series study. Journal of Experimental Orthopaedics. 2023;10:116. https://doi.org/10.1186/s40634-023-00668-y
Study Design: Retrospective single-centre case series. Level IV evidence. No control group; placebo effect cannot be excluded (authors cite Previtali et al. meta-analysis in discussion). Ethics committee approved (protocol 35/23 GAV, CET Lombardia 5). Authors note compliance with MIBO guidelines checklist as a study strength. Multilevel linear regression models used to account for bilateral treatment and adjust for gender, KL grade, and BMI.
Population: 49 patients (67 knees); 57.1% male. Mean age 57.7 (males), 61.1 (females). BMI 26.8–27.1. KL grade distribution: KL1 – 31.3%, KL2 – 61.2%, KL3 – 7.5%. All participants had failed ≥1 conservative treatment and ≥1 prior intra-articular injection (corticosteroid, PRP, or HA). Mean follow-up: 34.04 ± 13.62 months (range 11–59).
Intervention: Single intra-articular injection of 10 mL autologous MFAT processed using the Lipogems® system (Lipogems International Spa, Milan, Italy). Abdominal donor site; 13G blunt cannula harvest; closed-system mechanical micro-fragmentation without enzymatic treatment or culture expansion. No concurrent surgical procedures. Post-injection protocol: 5-day unloading; progressive weight-bearing; proprioceptive exercises from day 5.
Effect Sizes:
WOMAC (lower = better):
- Pain: 10.73 → 3.15 at 6 months (−70.6%); → 4.40 at 36 months (−59.0%); p<0.001 vs baseline at all timepoints
- Function: 34.76 → 11.21 at 6 months (−67.8%); → 15.36 at 36 months (−55.8%); p<0.001 vs baseline
- Total: 47.63 → 15.40 at 6 months (−67.7%); → 20.40 at 36 months (−57.2%); p<0.001 vs baseline
- Continued significant improvement from 3-month baseline to 6 months (p=0.016), 12 months (p=0.002), and 24 months (p=0.019)
KOOS Total (higher = better, 0–100 scale):
- Baseline: 38.45 ± 20.39
- 6 months: 71.59 ± 14.17 (+33.1 points; p<0.001)
- 12 months: 73.61 ± 17.72 (+35.2 points; p<0.001)
- 24 months: 73.29 ± 22.26 (+34.8 points; p<0.001)
- 36 months: 71.09 ± 25.69 (+32.6 points; p<0.001)
MCID Achievement – WOMAC (Holtz et al. thresholds: pain ≥7.5 pts, function ≥7.2 pts, total ≥10 pts):
- Function MCID: 76.1% / 79.1% / 81.5% / 83.3% / 80.0% at 3, 6, 12, 24, 36 months
- Pain MCID: 28.3% / 41.8% / 46.3% / 50.0% / 52.0% at 3, 6, 12, 24, 36 months
- Total MCID: 76.1% / 80.6% / 79.6% / 77.8% / 84.0% at 3, 6, 12, 24, 36 months
Prognostic factors (multilevel linear regression):
- Male gender associated with significantly worse WOMAC scores vs females: Δ14.3 pts at 3 months (p=0.017), Δ17.4 at 6 months (p=0.011), Δ16.3 at 12 months (p=0.043)
- KL grade 2 associated with significantly lower KOOS improvement vs KL grade 1 at 24 months: −26.2 points (p=0.042)
- Worse baseline WOMAC pain, function, and total scores significantly associated with greater likelihood of achieving MCID at all timepoints through 24 months (p<0.001 to p=0.034); association attenuated at 36 months (p=0.134–0.156)
Safety: Knee swelling and pain: 7 knees (10.4%), managed with cryotherapy, analgesics, and rest. Abdominal ecchymosis: 3 patients (6%), self-limited. No serious adverse events recorded. No patients required TKA at any point during follow-up.
Limitations:
- Level IV retrospective case series; no control group; placebo effect cannot be excluded
- Single centre; small sample (n=49 patients); wide follow-up range limits cross-timepoint comparability
- Late-timepoint sample sizes (n=36 at 24 months; n=25 at 36 months) reduce power and increase susceptibility to attrition bias
- Bilateral patients introduce data correlation (addressed via multilevel modelling, with sensitivity analyses excluding bilateral cases)
- Post-injection rehabilitation and analgesic use may independently contribute to functional improvement
- KL grade 4 patients eligible only if declining arthroplasty; none represented in published KL distribution (totals to 100% across KL1–3)
Learn More About Regenerative Matrix Therapy™ See More Conditions We Treat
Disclaimer
This content is intended for educational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any treatment decisions. Individual results vary based on disease severity, anatomy, and adherence to post-care protocols. Regenerative Matrix Therapy™ shares foundational similarities with, but is not identical to, the micro-fragmented adipose tissue technique studied in this research.
Why Choose Regenerative Matrix Therapy™ With RMIC?
RMIC delivers Regenerative Matrix Therapy™ through a standardized, physician-led protocol – ensuring consistent technique, appropriate patient selection, and structured care from initial consultation through recovery. Regenerative Matrix Therapy™ has been reviewed by Health Canada with no objection, reflecting the scientific rigor and safety framework underlying our approach. If you’re ready to explore whether you’re a candidate, we’re here to walk you through the process.